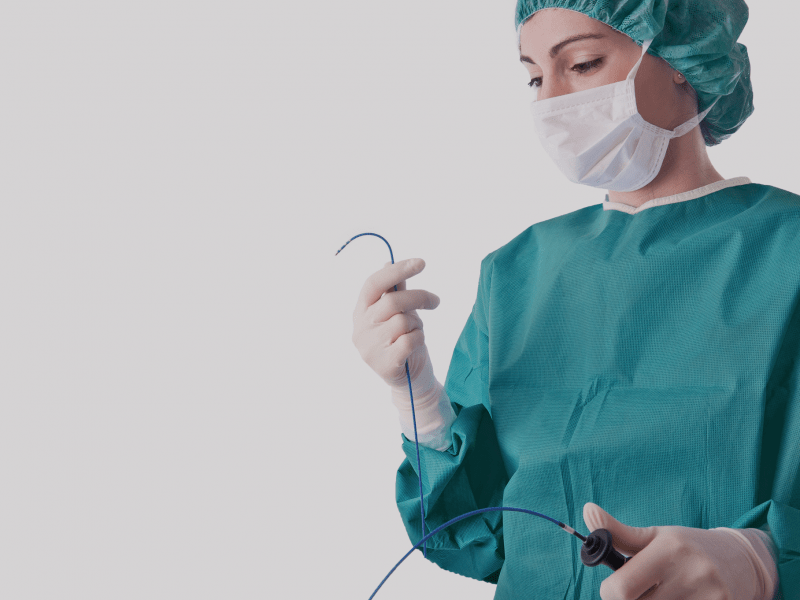
Electrophysiology devices
Vanguard has developed state of the art certified remanufacturing processes for more than 25 years. The remanufacturing of cooled ablation catheters and catheters with a 3D mapping function is one of our specialties.

CARTO3
Diagnostic and ablation catheters

EnSite Precision
Diagnostic and ablation catheters

More products
Discover our complete electrophysiology range
Vanguard sets the standard in medical remanufacturing of EP catheters
With the remanufacture of cooled ablation and 3D mapping catheters – Vanguard AG is taking medical remanufacturing of disposable medical products to a whole new level.
Treating atrial fibrillation
In atrial fibrillation, faulty electrical impulses throw the heart out of rhythm and the heart beats irregularly.
Coronary heart disease (atrial fibrillation) is the most common form of cardiac arrhythmia (irregular heartbeat) in adults worldwide. The lifetime risk for a 55-year-old European is between 35% and 40%. The risk of stroke is particularly high. In addition to increasing age, risk factors include (common) diseases such as high blood pressure, obesity, diabetes mellitus and frequent pauses in breathing at night.
In many cases, the occurrence of atrial fibrillation is triggered by extra electrical impulses from the pulmonary veins. This results in irregular electrical activity in the atria, which leads to an irregular and often too fast heartbeat. This leads to structural changes in the atria, which in turn favours the occurrence of atrial fibrillation.
If anticoagulant medication does not have the desired effect or is not an option, the heart can be successfully treated with catheter ablation, for example (see ESC guidelines, German Heart Foundation). Catheter ablation is now the treatment of choice for patients with atrial fibrillation who cannot be treated with medication to eliminate attacks or symptoms. The most important aim is to normalise electrical signals, restore the normal heart rhythm and thus prevent strokes.
With Vanguard’s restored catheters, the treatment of atrial fibrillation is sustainable, economical and safe.
The remanufactured medical devices from our electrophysiology portfolio meet all the needs of cardiac diagnostic and ablation procedures. You will not notice any difference between the remanufactured and original products when performing ablation procedures to treat abnormal heart rhythms. The patient will not feel the difference either.
Diagnosis and treatment of atrial fibrillation with 3D mapping
In minimally invasive catheter ablation, a catheter is (usually) advanced from the groin through the great vena cava to the heart.
The most commonly used method for isolating the pulmonary veins is point-by-point radiofrequency current ablation, which leads to cell necrosis through tissue heating. As the ablation catheter is controlled via electroanatomical mapping, this technique requires less radiation, but the examination is complex and can take longer. Another increasingly promising method is ablation with cold energy. The doctor scleroses a very specific area in the left atrium using high-frequency current or alternatively using cold (cryoballoon ablation). In both cases, this means that interference impulses can no longer reach the left atrium from this area – the area around the mouth of the four pulmonary veins – and trigger atrial fibrillation.
The success rate after one year with this method is 60 to 80 per cent, and around 90 per cent after a repeat procedure.
With the catheters for cardiac mapping systems, e.g. from Biosense Webster or Abbott, electrophysiologists can create a map of the heart, as with the original product, and thus obtain a clear picture of the electrical signals that control the heart rhythms. Once a map is created, doctors can identify electrical disturbances and areas of the heart that are causing heart rhythm problems and use ablation to treat the problem, such as atrial fibrillation. Cardiac ablation is a minimally invasive procedure to treat abnormally fast heartbeats. It involves creating lesions (ablations) in small areas of heart tissue that a doctor has identified as the cause of the arrhythmia.
Treatment options – Vanguard’s state of the art portfolio
Abbott’s EnSite Precision System or Biosense Webster’s Carto3 System have advanced imaging capabilities that enable real-time visualisation of a three-dimensional (3D) model of the patient’s cardiac anatomy. This allows physicians to more accurately identify areas of the heart that require ablation. The high-resolution cardiac mapping capability offered by these systems helps to quickly and accurately capture data and diagnose arrhythmias.
With Vanguard’s remanufactured catheters for the EnSite Precision System, physicians can opt for traditional impedance measurement (using mechanical cardiac activity) or electromagnetic technology, which enables precise localisation of the sensor-enabled catheters during treatment, as with the original product.
Biosense Webster is the industry leader in the diagnosis and treatment of cardiac arrhythmias. The Carto3 system is the most advanced three-dimensional imaging system for the electrophysiological treatment of cardiac arrhythmias. Even with the reprocessed catheters for the Carto3 system, the treatment of cardiac arrhythmias is just as fast and precise as with the original product, from the signals through diagnosis to a wide range of electrophysiological procedures.
Vanguard: Experts in their field
The biggest challenge in remanufacturing cooled ablation catheters lies in the cleaning of the long and narrow lumen of the catheter shaft so that no residue remains.
When remanufacturing catheters with 3D mapping systems – the electronic memory chips and magnetic field sensors require special attention.
We make make no compromises with regard to safety or functionality. Our cleaning machines and our testing equipment are the product of many years of research and development and are tailored to the specific requirements of remanufacturing complex medical devices.
Our catheters are subjected to more than 20 individual tests. These include:
Unique rinsing concept
Potential residues in the lumen are flushed out by a specially developed and patented cleaning machine. In addition, there is continuous volumetric monitoring of the media flowing through the lumen.

Optical inspection
Visual inspection, for e.g. curvature shape or damage starting from the electrical connection to the tip, with up to 40x magnification.

Mechanical function test
Checking of all moving components as well as the components for controlling the catheter. In the case of a catheter with a force measurement function its complete function is also checked.

Vanguard Product Verification (VPV)
Testing of the entire electrical functionality such as insulation, continuity, capacitance and the temperature sensors by a specially developed testing system.
Microbiological Testing
Ensuring the effectiveness of the cleaning process by testing for protein residues on each catheter using the modificated OPA method, reference method according to ISO 15883 and the in-house L2 hygiene laboratory all including documentation.
