Where is the Difference between Recycling and Remanufacturing a product?
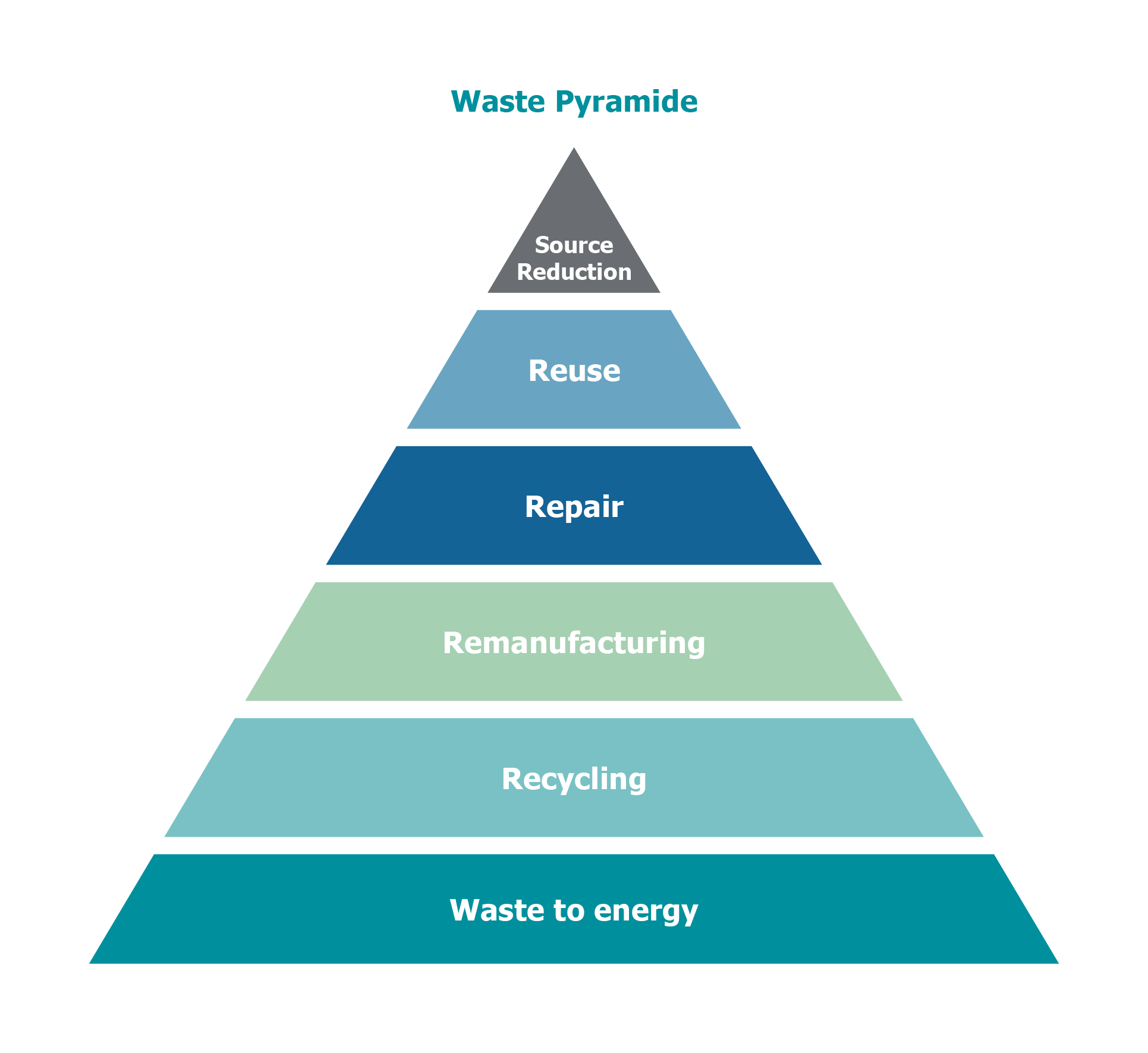
You may wonder if “recycled” and “remanufactured” can be used interchangeably, or if there are truly differences worth noting to understand how each contributes to environmental protection. In fact, the processes of remanufacturing and recycling fall into two distinct areas of the waste management hierarchy.
Medical Devices that have been remanufactured have been restored to their original functionality by replacing damaged or worn parts.
If that medical device were recycled instead, it would be dismantled, the hazardous materials would be removed, and the glass, plastic, precious metals and other recyclable contents would be converted back to raw materials for use in production of an entirely new product.
Reducing, reusing and recycling are all options for controlling our solid wastes. Remanufacturing the medical devices prevents waste by reusing it, while recycling manages the wastes created when the medical devicer is discarded.
Recycling and remanufacturing also differ in the environmental benefits achieved by each process. Both methods reduce solid and hazardous wastes compared to landfilling, but remanufacturing outshines recycling in the reduction of energy use, greenhouse gasses, and air and water emissions. These differences can largely be attributed to the additional handling and transportation required for reintroducing recycled materials into the manufacturing process.
Recycled-content products are widely available and cover many categories of consumer goods, from paper and other school supplies to clothing and furniture. These items may be 100 percent recycled, where no virgin materials were used, or just a portion of the materials may have been recycled.
Remanufactured products, on the other hand, are more limited in nature, but include a range of items, from automotive parts to furniture, tires to toner cartridges, and computers to medical equipment.
We at Vanguard have set ourselves the goal of enabling and strengthening the circular economy within the medical technology and healthcare sector. Join us!





